Thinking about one of our Professional MS in Pharmaceutical Sciences programs? Check out our resources to determine which program is the best one for you.
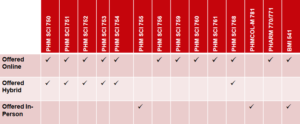
While the PPI M.S. is offered in a fully online format, the ADD M.S. can either be completed fully online, or as a hybrid program, with both online and in-person components. None of our programs can be completed fully in-person, as some required courses are only offered online.
Similarly, these programs differ from the Pharmaceutical Sciences PhD program; they are not research-focused, and coursework for the PPI or ADD programs will not count towards the Pharmaceutical Sciences PhD.
The Programs: ADD vs PPI
| Applied Drug Development |
Psychoactive Pharmaceutical Investigation
|
|
| Applicant Degree Requirements | Bachelor’s degree in STEM field |
Bachelor’s degree with
strong science background
|
| Time to Degree | 1 year (accelerated), 2-3 years (part-time) |
1 year (accelerated),
2-3 years (part-time)
|
| Student Funding | Self-funded, limited scholarships exist |
Self-funded, limited
scholarships exist
|
| Type of Experiential Learning | Internship |
Internship or
Research Course
(elective options)
|
| Types of Hiring Companies and Agencies |
|
|
| Example Job Titles |
|
|
Fully Online (PPI or ADD)
Complete the program entirely online. This program’s classes are mostly asynchronous to provide greater flexibility. Take this program in any location and time zone.Typical students who choose this option:
- Working professionals
- Students located outside of Madison
Hybrid (ADD only)
Complete the program through a combination of online, hybrid, and in-person classes. This program includes on-campus meetings such as labs as well as some classes that are entirely online.Typical students who choose this option:
- International students seeking visa
- Students seeking one hands-on lab course
Breakdown of each course modality
| Most learning activities are provided, completed, and submitted | Expected live class sessions | Interactions with Instructors | |
| Online Sections | Online | Online 3-6 times per semester | Online |
| Hybrid Sections | Online | On campus 3-6 times per semester | Online |
| In-Person Sections | In-person on campus | Scheduled in-person on campus meetings | In-person on campus |
Course Type Descriptions
- Online: All course materials are provided and all activities are completed through online course software. Most of the coursework is completed asynchronously, but there may be selected synchronous class meetings held on virtual platforms throughout the semester.
- Hybrid: Most course materials are provided and most activities are completed through online course software. Much of the coursework is completed asynchronously, with select in-person synchronous class meetings held throughout the semester.
- In-Person: Instruction is provided on the UW-Madison campus. Students must be able to attend all class meetings in person
These programs are terminal MS degrees that differ from the Pharmaceutical Sciences PhD program; they are not research-focused, and coursework for the PPI or ADD programs will not count towards the Pharmaceutical Sciences PhD.
| ADD/PPI MS (Non-research degree program) | Pharmaceutical Sciences PhD | |
| Focus | Professional preparation for industry work | Academic research |
| Course Format | Mostly to fully online | On-campus and in-person |
| F-1 Visa Eligibility | Yes, for ADD option, with enrollment in specific courses taken at accelerated rate. PPI is not eligible for Visa. | Yes |
| Instructors | Industry experts (many do not work on campus) | On-campus research faculty |
| Time to Degree | 12-16 months accelerated (needed for visa status) or 2-3 years part-time | ~5 years |
| Student Funding | Self-funded, limited scholarships exist; not eligible for funded assistantships | Assistantships for teaching/research |
| Type of Experiential Learning | Industry internship | Independent research |
| Types of Hiring Companies and Agencies | Drug development companies, contract research organizations, drug manufacturers, regulatory agencies | Academia, pharmaceutical industry, scientific communications, fellowships |
| Example Job Titles | Research Associate, Quality Assurance Associate, Clinical Affairs Associate, Regulatory Affairs Specialist | Professor, Post-Doctoral Fellow, Scientist/Senior Scientist, Strategic Marketing Specialist, Scientific Communications Associate/Editor |
